Thrombin-like enzyme
Thrombin is a trypsin-like serine protease protein that acts in the blood coagulation cascade. It converts soluble fibrinogen into insoluble strands of fibrin, as well as catalyzing many other coagulation-related reactions.
Snake venom thrombin-like enzymes (svTLEs) are serine proteases that are, like thrombin, able to clot fibrinogen and play an important role in the process of blood coagulation. However, unlike thrombin, svTLEs cannot activate factor XIII, which is essential in blood-clot stabilization. Consequently, blood clots produced by SVTLEs are not stable and are cleared rapidly.
svTLEs are divided into three groups, depending on which fibrinopeptide (FP) they cleave off, FPA from fibrinogen Aα-chain (FGA), FPB from fibrinogen Bβ-chain (FGB) or both. They commonly cleave off either FPA or FPB, but rarely both fibrinopeptides, as does thrombin.
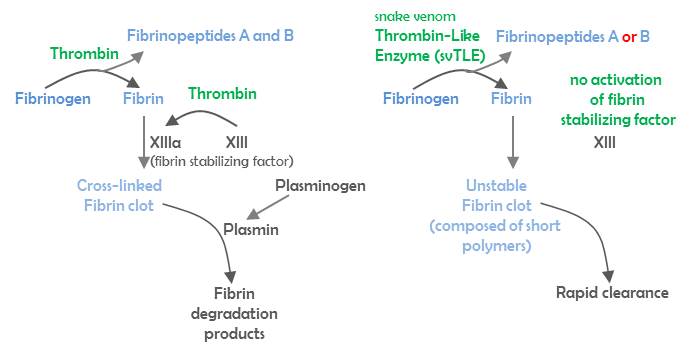
Figure: The last stops of blood coagulation cascade and fibrinolysis in physiological conditions (left panel) and in the presence of snake venom thrombin-like enzyme (svTLE) (right panel).
